News
Welcome to our news section. Here you can keep up to date with the latest news from GMP+ International regarding the GMP+ FC scheme, feed market news and more.
Last update
GMP+ International publishes annual report 2024
Our annual report 2024 is now available. The report marks a year of change and strengthening, with the introduction of a new vision, mission and core values, as well as the appointment of our new Managing Director, Martine Boon.

GMP+ International Partners with AGER: A First in Italy
We are glad to announce GMP+ International’s first official partnership in Italy with AGER (Emilian-Romagna Granary Association), the well-respected trader ...
Published: 24 June 2025

Trusted partnership continues: GMP+ & Farm Dairy Code
GMP+ International has reaffirmed its long partnership with Bond van Boerderij-Zuivelbereiders (BBZ), ensuring the safe integration of farm cheese whey ...
Published: 24 June 2025

Extension of Supply Chain Model Recognition Responsible Soy Until end of 2026
Continued Recognition of Supply Chain Models Book & Claim and Mass Balance.
Published: 24 June 2025

Improved Prerequisites Course Group for Safer Feed
Getting the basics right is essential before applying HACCP principles. That’s why we have updated our Prerequisites Course Group on ...
Published: 24 June 2025
International Database for Transport of Feed (IDTF) – Newsletter 2/2025
To ensure continuous updating of the IDTF database, the International Committee for Road Transport (GMP+ International, OVOCOM, QS, Oqualim, EFISC-GTP, ...
Published: 17 June 2025

TopCrop fully complies with GMP+ FC requirements
TopCrop (formerly VVAK) has been a reliable supplier of unprocessed agricultural products for animal feed production for many years.
Published: 10 June 2025

Discover the new GMP+ Academy brochure
We are excited to share the brand-new GMP+ Academy Brochure. This digital brochure provides a clear introduction to the GMP+ ...
Published: 27 May 2025

New Registered Consultant in Spain – Welcome, CENTAC Group
With a strong mix of practical experience and strategic insight, CENTAC group supports companies in Spain to streamline processes, enhance ...
Published: 27 May 2025

Invalid certificates issued
We would like to inform you of a recently reported case involving invalid certificates issued in the name of Transportadora ...
Published: 15 May 2025

Invalid certificates issued
We would like to inform you of a recently reported case involving invalid certificates issued in the name of Nortem ...
Published: 8 May 2025

Explore the New GMP+ platform – Updates, Features, and Improved Usability
As of January 2025, the entire GMP+ Community has access to our fully updated digital platform. Whether you are a ...
Published: 29 April 2025
Knowledge is the foundation of safe and sustainable feed
At GMP+ International, we believe that certification is not the starting point it’s the final step in a much broader ...
Published: 29 April 2025

New: Explanation of GMP+ certification tariffs now on our website
We're committed to supporting companies on their journey toward feed safety and sustainability.
Published: 29 April 2025

Updated risk assessments in Risk Management Tools
Discover the value of the Product list and (new) feed materials accepted by our Technical Committee Risk Management
Published: 28 April 2025
Protocol Monitoring Aflatoxin B1 in maize and by-products of maize
Change risk profile for USA. As a result of the evaluation of the available analysis results of Aflatoxin B1 in maize ...
Published: 24 April 2025
Protocol Monitoring Aflatoxin B1 in maize and by-products of maize
Change risk profile for Slovakia As a result of the evaluation of the available analysis results of Aflatoxin B1 in maize ...
Published: 28 March 2025

GMP+ and Trans.EU partner up
GMP+ International has partnered with Trans.eu, owner of an advanced transport management platform. This collaboration aims to integrate the Transport ...
Published: 25 March 2025

GMP+ Academy partners with Collaborative Soy Initiative
We are pleased to announce our new partnership with the Collaborative Soy Initiative (CSI).
Published: 25 March 2025

Global Trusted Auditors supporting feed companies in Brazil as a GMP+ Registered Consultant
Global Trusted Auditors (GTA), a consultancy company based in Brazil, is now officially a GMP+ Registered Consultant! This means, they ...
Published: 25 March 2025

Harmonisation Meeting in Kuala Lumpur – A great success!
On 20 February 2025, we came together in Kuala Lumpur for a Harmonisation Meeting with the goal of optimising feed ...
Published: 25 March 2025

International Database for Transport of Feed (IDTF) – Newsletter 1/2025
To ensure continuous updating of the IDTF database, the International Committee for Road Transport (GMP+ International, OVOCOM, QS, Qualimat, EFISC-GTP, ...
Published: 6 March 2025

PRESS RELEASE - Feed safety scheme holders in Europe, unite to harmonise
Agreement on the prolongation of the mutual recognition between CSA-GTP and EFISC-GTP, GMP+ International and OVOCOM
Published: 5 March 2025

GMP+ Feed Certification scheme changes
We are pleased to announce that the changes to the GMP+ FC scheme documents have been published. In collaboration with ...
Published: 3 March 2025

Protocol Monitoring Aflatoxin B1 in maize and by-products of maize
Change risk profile for Austria and USA As a result of the evaluation of the available analysis results of Aflatoxin B1 ...
Published: 28 February 2025

Upcoming GMP+ Feed Certifications scheme changes – Effective from 3 March 2025
We're pleased to inform you about the key changes to the GMP+ Feed Certification scheme! In collaboration with our Subcommittees ...
Published: 27 February 2025

Consilium | food & feed joins GMP+ International as a Registered Consultant in Poland
GMP+ International welcomes Consilium | food & feed as a new GMP+ Registered Consultant and a GMP+ Academy Registered Trainer ...
Published: 27 February 2025

Important Update: New Terms of Use effective from 11 February 2025
As of 11 February 2025, the updated Terms of Use for the GMP+ Platform will come into effect. This means ...
Published: 6 February 2025
Coming in March 2025 - GMP+ TS2.2 Antibiotic-Free Feed as a global standard
The GMP+ TS2.2 Antibiotic-Free Feed standard is evolving into a global standard starting in 2025. This standard empowers feed producers ...
Published: 28 January 2025

Now Available: European Sustainability Legislation Updates on FeedLegislation.org
Starting in January 2025, you can stay informed about European sustainability legislation affecting the feed sector through the Feed Legislation ...
Published: 28 January 2025

Prerequisites: Most popular in 2024!
In the feed industry, it is essential that companies have their prerequisites in place. This is referred to internationally as ...
Published: 28 January 2025
Our new platform is live! Explore it now
Hooray! Our new GMP+ platform is live! We'd love to show you some highlights! Here is what to expect:
Published: 21 January 2025

Executive decree: extended by 12 months until 31 December 2025
This executive decree on the GMP+ transport of feed products inside the Ukraine and from Ukraine to other countries, has ...
Published: 20 December 2024

GMP+ FC scheme: Important update on publication schedule
We’re introducing a new publication schedule for the GMP+ Feed Certification (FC) scheme changes. Starting in 2025, the updated versions ...
Published: 18 December 2024

Our customers and partners value our products and services
As part of our three-year corporate plan, we set ourselves the goals of continuously increasing the added value of the ...
Published: 18 December 2024

NEW: European sustainability legislation on the Feed Legislation website
GMP+ International is committed to safe and sustainable feed, worldwide. Every company that is active in the feed industry must ...
Published: 18 December 2024

Webinar on supplier assessment & management
Last month, we had the privilege of interviewing Anne Mette Onarheim from Nutreco, who has over 30 years of experience ...
Published: 18 December 2024

Contamination with Aflatoxin B1 in maize originating from Hungary or Slovakia
Contamination with Aflatoxin B1 is observed in maize gluten feed, produced from maize originating from Hungary or Slovakia. Be ...
Published: 17 December 2024

Update risk assessments in risk management tools
Within the GMP+ Feed Certification scheme (GMP+ FC scheme), feed materials and their processes must be subjected to a safety ...
Published: 12 December 2024

International Database for Transport of Feed (IDTF) – Newsletter 3/2024
To ensure continuous updating of the IDTF database, the International Committee for Road Transport (GMP+ International, OVOCOM, QS, Qualimat, EFISC-GTP, ...
Published: 5 December 2024

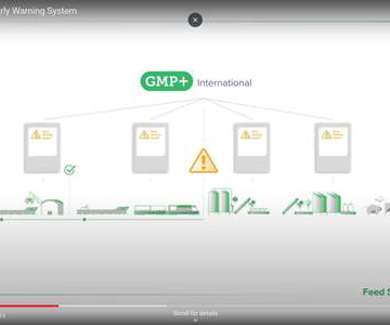
Changing the EWS emergency number – please update your documentation
Please note that the emergency number for the Early Warning System (EWS) has changed. The Early Warning System (EWS) plays a ...
Published: 28 November 2024

Have you explored our learning platform, The GMP+ Academy, yet?
It is designed to provide feed professionals like you with resources to gain knowledge and skills on feed safety and ...
Published: 28 November 2024

GMP+ International partners with Samen Tegen Voedselverspilling for a safer, more sustainable feed supply chain
GMP+ International partners with Samen Tegen Voedselverspilling to tackle food waste and enhance feed safety. This partnership unites our efforts ...
Published: 28 November 2024

Early Warning System Notifications Report 2023
GMP+ International received 10% more Early Warning System (EWS) notifications in 2023 compared to 2022. This increase may be attributed ...
Published: 28 November 2024

Results from GMP+ Monitoring database now available
Do you want more insight for your feed safety management system? We have made an infographic of all the data ...
Published: 28 November 2024

Protocol Monitoring Aflatoxin B1 in maize and by-products of maize
Change risk profile for Austria and Czech Republic. As a result of the evaluation of the available analysis results of Aflatoxin ...
Published: 21 November 2024

Tariffs GMP+ International 2025
As of 1 January 2025, GMP+ International will adjust the tariffs for the contributions of the Certification Bodies and of ...
Published: 11 November 2024

New GMP+ numbers mandatory from 1 December 2024
As of 1 December 2024, the transition period of 5 years will expire and the old GMP+ registration numbers can ...
Published: 31 October 2024

Coming soon, e-learning on Supplier Assessment & Management!
The GMP+ Academy will be launching two new e-learning modules on supplier assessment and management on 7 November 2024.
Published: 31 October 2024

Join our exclusive webinar on Supplier Assessment & Supplier Management!
Are you looking to strengthen your supplier relationships and ensure quality? Don’t miss this opportunity to hear directly from Anne ...
Published: 31 October 2024

Going live soon: a new login environment
We would like to inform you of some important developments at GMP+ International to further increase the added value ...
Published: 30 September 2024
Survey invite: Your chance to give feedback to GMP+ International
Together with independent market research company Ipsos, we are inviting you to give feedback on the GMP+ FC scheme and ...
Published: 30 September 2024

Introducing our new course group: Basic Feed Safety Control
Feed safety is very important in feed production. Is your team ready to identify risks, create a monitoring plan, and ...
Published: 30 September 2024

The collaborative efforts of PUM and GMP+ International
GMP+ International and PUM are excited to announce a strategic collaboration aimed at enhancing feed safety standards in emerging markets. ...
Published: 30 September 2024

Do you produce and/or trade soy?
The certification deadline for FRA Responsible Soy Standards is 31-12-2024 for specific markets.
Published: 24 September 2024

Protocol Monitoring Aflatoxin B1 in maize and by-products of maize - Harvest 2024/2025
Change risk profile for Czech Republic and Romania. As a result of the evaluation of the available analysis results of Aflatoxin ...
Published: 12 September 2024

Updated version of the TS1.7 – Monitoring now available
Due to legislation and sector changes, GMP+ International, in collaboration with the International Expert Committee (IEC), has updated the TS1.7 ...
Published: 2 September 2024

Martine Boon announced as incoming GMP+ International Managing Director
Feed Safety first, building on what has been achieved, and expanding sustainability support for companies high on her agenda.
Published: 27 August 2024

EC Consulting in Indonesia joins as a GMP+ Registered Consultant & Academy partner
GMP+ International welcomes EC Consulting, a leading firm in food safety solutions across various sectors in Indonesia as our newest ...
Published: 30 July 2024

GMP+ International, ALL4Feed and RTRS join forces at SPACE 2024
We are excited to announce that GMP+ International, ALL4Feed and the Round Table on Responsible Soy (RTRS) will exhibit together ...
Published: 30 July 2024

Celebrate with Us: 3 Years of GMP+ Academy!
To celebrate the 3rd anniversary of the GMP+ Academy, we are giving away 3 sampling e-learning modules.
Published: 30 July 2024

Annual Harmonisation Training for GMP+ Registered Consultants
The annual harmonisation training for GMP+ Registered Consultants brought together over 30 consultants worldwide. This year the focus was on ...
Published: 30 July 2024

Update risk assessments in risk management tools
Within the GMP+ Feed Certification scheme (GMP+ FC scheme), feed materials and their processes must be subjected to a safety ...
Published: 29 July 2024

Executive decree: extended by six months until 31 December 2024
This executive decree on the GMP+ transport of feed products inside the Ukraine and from Ukraine to other countries, has ...
Published: 18 July 2024

International Database for Transport of Feed (IDTF) – Newsletter 2/2024
To ensure continuous updating of the IDTF database, the International Committee for Road Transport (GMP+ International, OVOCOM, QS, Qualimat, EFISC-GTP, ...
Published: 3 July 2024

Translate our website in your own language!
We're going to learn about browser translation tools. These tools help you read our websites in your own language. This ...
Published: 28 June 2024
Introducing Wenxin, our new Helpdesk colleague
If you contact the GMP+ Helpdesk soon, you may find yourself speaking to our new colleague Wenxin, who joined GMP+ ...
Published: 27 June 2024

New GMP+ International website
Have you seen our new website yet? As of April 2024, our new website has been successfully launched. We’ve received ...
Published: 27 June 2024
New e-learning module on Sampling for the Feed Industry
The GMP+ Academy is launching an e-learning module on sampling. Every feed company needs to sample its ingredients and products ...
Published: 27 June 2024

Protocol Monitoring Aflatoxin B1 in maize and by-products of maize
Change risk profile for Hungary. As a result of the evaluation of the available analysis results of Aflatoxin B1 in maize ...
Published: 7 June 2024

Meet our new partner Apheya Animal Nutrition
Introducing our latest partner in the United Kingdom: Apheya Animal Nutrition. They bring extensive knowledge in feed production, safety protocols, ...
Published: 30 May 2024

The most frequently asked questions about the new website
A month after going live, we are now taking stock. A new website with a number of new features - ...
Published: 30 May 2024

Have you already seen the new GMP+ website?
Our updated website is live. After extensive research focusing especially on listening to your wishes, this is the result!
Published: 30 April 2024

Update risk assessments in FSP
Within the GMP+ Feed Certification scheme (GMP+ FC scheme), feed materials and their processes must be subjected to a safety ...
Published: 25 April 2024

First company complies with GMP+ MI5.5 Carbon footprint of feed
In March, Agrifirm, based in Apeldoorn, The Netherlands, became the first company to receive a statement of compliance with the ...
Published: 10 April 2024

Protocol Monitoring Aflatoxin B1 in maize and by-products of maize
Change risk profile for Czech Republic, Romania and Serbia. As a result of the evaluation of the available analysis results of ...
Published: 8 April 2024

First GMP+ Registered Consultant in Romania: Welcome TQ Services
“Being a GMP + Registered Consultant is a recognition of the company’s capabilities of providing professional services.” TQS Manager and ...
Published: 28 March 2024

Introducing HTP Advies: GMP+ Registered Consultant and Training Institute for the GMP+ Academy
HTP Advies has deep expertise in setting up systems for the animal feed and food industries, covering production, storage, transportation, ...
Published: 28 March 2024

New website almost live
After months of hard work, our new website is about to go live at the end of April. After extensive ...
Published: 28 March 2024
Share your analysis results in the GMP+ Monitoring database.
Every year, GMP+ International evaluates the data in the GMP+ monitoring database. Before we start with the evaluation of 2023, we ...
Published: 28 March 2024

Protocol Monitoring Aflatoxin B1 in maize and by-products of maize
Change risk profile for Ukraine As a result of the evaluation of the available analysis results of Aflatoxin B1 in maize ...
Published: 8 March 2024

A message from GMP+ International Managing Director, Roland van der Post
After careful consideration and reflection on my journey with GMP+ International I have decided that the time has come for ...
Published: 5 March 2024

GMP+ International and RTRS renew their collaboration agreement
GMP+ International and the Round Table on Responsible Soy have redefined their collaboration. In a significant move towards promoting the use ...
Published: 28 February 2024

Launch of Refreshed HACCP E-Learning!
In early February, we launched our refreshed HACCP e-learning. The feedback we have received from the community has been valuable, ...
Published: 28 February 2024

A new partnership with Akademie Burg Warberg
We are proud to announce that Akademie Burg Warberg is now a partner of the GMP+ Academy. This renowned German institute ...
Published: 28 February 2024

Our Newest Registered Consultant from Spain: Zitec Consultores
A new Registered Consultant in Spain. Zitec Consultores provides knowledge and experience to help us achieve feed safety in Spain.
Published: 28 February 2024

Brochure training offering
Start the new year right—invest in yourself. Explore the updated brochure and choose a (online) training that fits your goals. ...
Published: 14 February 2024

International Database for Transport of Feed (IDTF) – Newsletter 1/2024
There have been several changes for the IDTF by the International Committee for Road Transport (GMP+ International, OVOCOM, QS, Qualimat, ...
Published: 8 February 2024

Protocol Monitoring Aflatoxin B1 in maize and by-products of maize
Change risk profile for Ukraine and SW France As a result of the evaluation of the available analysis results of Aflatoxin ...
Published: 1 February 2024

Successful FRA-trainings for Certification Bodies
On December 19 and 21, 2023, GMP+ International conducted successful training sessions on the GMP+ FRA module and associated standards ...
Published: 30 January 2024

Our customers and partners value our products and services
As part of our three-year corporate plan, we set ourselves the goals of continuously increasing the added value of the ...
Published: 29 December 2023

Important update: new AMA Quality Seal requirements regarding soy from January 1, 2024
For companies that produce and/or trade soybeans, soy products and/or compound feed containing soy for the AMA Quality Seal
Published: 28 December 2023

Important update for companies with QM-Milch Scope
As of 01-01-2024, there are important changes to the conditions for companies that supply dairy feed for the dairy farmers ...
Published: 22 December 2023

Updates in the GMP+ FSA and new FRA documents have been released
The improvements in the documents of the GMP+ Feed Certification scheme and two new normative documents are now available our ...
Published: 15 December 2023

Update risk assessments in FSP
Within the GMP+ Feed Certification scheme (GMP+ FC scheme), feed materials and their processes must be subjected to a safety ...
Published: 14 December 2023

Important updates for companies: new AMA Quality Seal requirements regarding soy from January 1, 2024
For companies that produce and/or trade soybeans, soy products and/or compound feed containing soy for the AMA Quality Seal. As ...
Published: 6 December 2023

Tariffs GMP+ International 2024
As of 1 January 2024, GMP+ International will adjust the tariffs for the contributions of the Certification Bodies and of ...
Published: 30 November 2023

1000 companies certified for GMP+ FRA and two new standards on the way
At a time when sustainability is a topic for discussion throughout society, the feed industry is taking a step forwards. ...
Published: 30 November 2023

Users give GMP+ Academy 8.3 out of 10!
Good news – the GMP+ Academy has earned a rating of 8.3 out of 10!
Published: 30 November 2023
Carbon footprint standard for the Dutch market on the way
With GMP+ Feed Responsibility Assurance (GMP+ FRA) certification, companies are demonstrating that they are complying with the market requirements for ...
Published: 31 October 2023

Which news would you like to receive?
At GMP+ International we believe that sharing relevant news and knowledge with our Community is a prerequisite for a safe ...
Published: 30 October 2023

Welcome Control Union Argentina
It is with great pleasure that we introduce our latest partner, Control Union Argentina, as a GMP+ Registered Service
Published: 30 October 2023

International Database for Transport of Feed (IDTF) – Newsletter 2/2023
To continuously improve the IDTF database and to achieve greater coherence between IDTF-numbers for products covered by Regulation (EC) No. ...
Published: 30 October 2023

Sustainability, the next steps
In today’s changing world, our sector needs to step up to the challenge of producing and operating in a sustainable ...
Published: 27 September 2023

Our customers and partners value our products and services
As part of our three-year corporate plan, we set ourselves the goals of continuously increasing the added value of the ...
Published: 26 September 2023

A changed market calls for a new soya standard
At GMP+ International, we believe that deforestation and its impact on sustainability is an important issue.
Published: 26 September 2023

New GMP+ FSA & GMP+ FRA logos now available to download
The new GMP+ FSA and GMP+ FRA logos are now available. You can use the logos as described in F0.1 ...
Published: 26 September 2023

Improvements & developments in our services
As part of our corporate strategy and based on customer research, we aim to increase the added value of our ...
Published: 26 September 2023

Infographic results from GMP+ Monitoring database now available
Do you want to have more insights for your feed safety management system? We have made an infographic of all the ...
Published: 31 July 2023

GMP+ FRA Responsible soy standards compliant with requirements of QS
The GMP+ FRA standards for the production and trade of responsible soy will be recognized for the QS Add-on module: ...
Published: 31 July 2023

Protocol Monitoring Aflatoxin B1 in maize and by-products of maize
Change risk profile for Canada As a result of the evaluation of the available analysis results of Aflatoxin B1 in maize ...
Published: 26 July 2023

Update risk assessments in FSP
Within the GMP+ Feed Certification scheme (GMP+ FC scheme), feed materials and their processes must be subjected to a safety ...
Published: 13 July 2023

Free Microlearning Module “What are prerequisites?”
Prerequisites are the foundation of the HACCP system. Learn more about hygiene practices that represent the basic conditions and the activities ...
Published: 29 June 2023

QESH Consult ApS becomes first GMP+ Registered Consultant in Denmark
“Our primary goal is to help the company develop in a direction where there is more focus on feed safety ...
Published: 29 June 2023

GMP+ International Annual Report 2022 now available
Our annual report contains our corporate achievements and our financial results for 2022 and illustrates the ways in which we ...
Published: 29 June 2023

Early warning of mycotoxins in European cereal crops
GMP+ International is working together with Wageningen Food Safety Research, SGS, Cargill, Alltech and the Committee of Grain Traders to ...
Published: 29 June 2023

Executive decree: extended by twelve months until 30 June 2024
This executive degree on the GMP+ transport of feed products inside the Ukraine and from Ukraine to other countries, has ...
Published: 29 June 2023

Protocol Monitoring Aflatoxin B1 in maize by-products
Change risk profile for Canada, Italy, Ukraine and Southern France. As a result of the evaluation of the available analysis ...
Published: 23 June 2023

International Database for Transport of Feed (IDTF) – Newsletter 1/2023
Several changes for the IDTF database
Published: 6 June 2023

Welcome Qualikadi - a new Registered Consultant & Registered Training Institute
“Our mission is to promote the knowledge development of people and organisations in the feed and food sector, helping them ...
Published: 30 May 2023

How to use the GMP+ Monitoring database?
Instructional video now available
Published: 30 May 2023

Lesson learned: requirements are not always implemented correctly
Following the recent EWS notifications on Aflatoxin B1 in feed we found that unclear expectations and unfamiliarity with some requirements ...
Published: 28 April 2023

Now available: video on how to submit an EWS notification
A GMP+ certified company must ensure that the supplied feed is safe. Still things can go wrong, in that case ...
Published: 28 April 2023
New e-learning: “Principles of the GMP+ Feed Certification scheme"
A number of incidents in the late 80s and 90s shook the market. They were part of the reason why ...
Published: 28 April 2023

Update risk assessments in FSP
Within the GMP+ Feed Certification scheme (GMP+ FC scheme), feed materials and their processes must be subjected to a safety ...
Published: 21 April 2023

Protocol Monitoring Aflatoxin B1 in maize by-products
Change risk profile for Ukraine As a result of the evaluation of the available analysis results of Aflatoxin B1 in maize ...
Published: 13 April 2023

Upload your analysis results
Share your analysis results in the GMP+ Monitoring database. Every year, GMP+ International publishes an infographic for the GMP+ Community showing ...
Published: 30 March 2023

ABIOVE new partner for GMP+ International
GMP+ International focuses on the importance of feed safety in the entire feed supply chain. ABIOVE and GMP+ International will ...
Published: 30 March 2023

Increase your knowledge about HACCP in less than 20 minutes
Feed certification schemes are about risk management in terms of feed safety. Hazard Analysis & Critical Control Points (HACCP) is ...
Published: 30 March 2023

Developments and improvement of GMP+ services
Earlier this month we started a long-term partnership with two companies, Xuntos & Online Department, to improve our services for ...
Published: 30 March 2023

Are you ready for the transition to the GMP+ FC scheme 2020?
Certification has been possible under the requirements of the GMP+ Feed Certification (GMP+ FC) scheme 2020 for some time. A ...
Published: 28 February 2023

Risk assessments; how can they help you?
Via the Feed Support Products (FSP), we share valuable information with the GMP+ certified companies. These products help you manage ...
Published: 28 February 2023
Protocol Monitoring Aflatoxin B1 in maize by-products
Change risk profile for Czech Republic As a result of the evaluation of the available analysis results of Aflatoxin B1 in ...
Published: 27 February 2023

Good news, scheme documents now published in 8 languages
Updates to the requirements for several scheme documents about FSA are now published on our website in 8 languages.
Published: 30 January 2023

The GMP+ Helpdesk
Get to know our colleagues from the GMP+ Helpdesk.
Published: 30 January 2023

New GMP+ Registered Consultant and Registered Training Institute from Brazil, welcome!
Practical and theoretical knowledge are the main tools for understanding a feed safety management system. The growing number of GMP+ ...
Published: 30 January 2023

CKP launches a microlearning on feed safety culture
The Collective Knowledge Programme (CKP) has developed a series of seven short microlearning modules
Published: 10 January 2023

